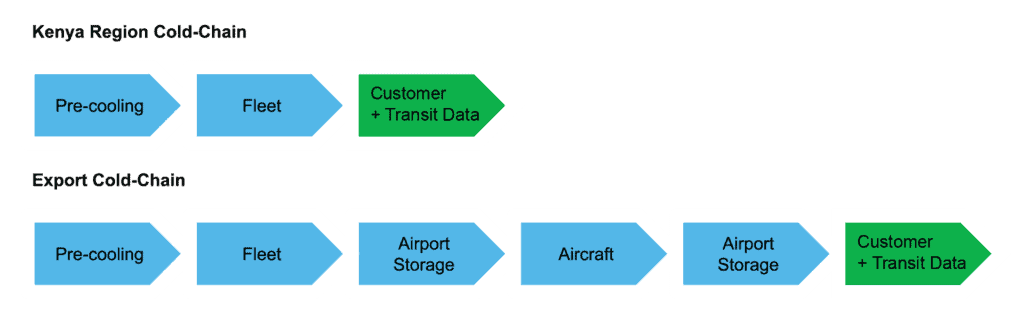
The effect of Beauvitech on nature’s little helpers
By John Ogechah and Cory Smit

Beauveria bassiana is a well-known soil-dwelling entomopathogenic (insect-killing) fungus found all over the world. For more than 100 years, tons of B. bassiana spores (e.g. Beauvitech®) have been commercially produced and used for biological control of insect pests worldwide. Growers have come to rely on this clever biological action known as white muscardine disease as a major player in chemical-free pest control.
Following increased interest in biocontrol of pest insects between 1980 and 1990, safety aspects were raised and discussed in great detail. Burges (1981) outlined the main principles and guidelines for testing the safety of insect pathogens and “that a pathogen should be registered as safe when there is reasonable evidence that it is so and in the absence of concrete evidence that it is not. A “no risk” situation does not exist, certainly not with chemical pesticides, and even with biological agents one cannot absolutely prove a negative.”
The commercial use of entomopathogenic fungi and their products as mycoinsecticides (fungus-based insecticides), therefore, necessitates their registration based on certain safety guidelines. Beauveria bassiana is indeed registered in several countries and proof of safety to non-target organisms such as mammals, fish, amphibia, birds, pollinators etc is an important requirement before registration.
Still, the lingering question posed by farmers and indeed the greater society is “What about the bees?” In other words, how safe are mycoinsecticides and, specifically, Beauveria bassiana to these little helpers of nature?
There are numerous peer-reviewed papers on the effect of B. bassiana on honeybees and other beneficial organisms. Examples are presented in Table I below. Notable is the fact that most of the studies were done in the laboratory and only a few in the field.
The vast majority of the studies done on bees conclude that despite the wide host range of B. bassiana, this fungus can be used with minimal impact on honeybees and other non-target organisms. Some experiments showed that B. Bassiana can be blown directly into hives to manage Varroa destructor mites (Acari: Varroidae) without a negative effect on the bee colonies (Miekle et al., 2008; Rodríguez et al. 2009). Another set of experiments looked at using honeybees to distribute B. bassiana spores directly to crop flowers and foliage (Almazra’awi et al. 2006). Similarly, no adverse effects on the bees were reported.
In one case, however, Almazra’awi (2007) reports that B. bassiana strains caused high mortality in caged bees dusted with dry formulations of high concentrations (10⁸-10⁹ CFUg-1). Interestingly, in the same paper (Almazra’awi, 2007), exposure of whole beehives under field conditions resulted in low mortality that was not different from the controls regardless of the isolate tested. This points to the difference between the physiological host range and the ecological host range (Hajek & Butler 2000).
The physiological host range demonstrates the range of insect species that can be infected in the laboratory, while the ecological host range demonstrates which insects can be infected in nature or under field conditions. Non-target insects which are infected under laboratory conditions, may not necessarily be infected in nature (Zimmermann, 2007).
We conclude that despite the wide host range of B. bassiana, evidence to date suggests that this fungus can be used with minimal impact on non-target organisms, especially when isolate selection and spacio-temporal factors are taken into consideration. Our answer is unwavering: Beauveria bassiana (Beauvitech®) has no negative effect on honeybees (Apis mellifera) in normal field conditions. In fact, there are numerous examples of benefits B. bassiana can have with and for bees.
|
Table I. Examples of effects of B. bassiana (strains and formulations) on beneficial and non-target organisms.
|
| Beneficial organism |
Fungus (Strain/ Formulation) |
Lab./ Field Trials (L/F) |
Results/Observations |
Reference |
| Amblyseius cucumeris |
B. bassiana (Naturalis-L, BotaniGard WP) |
L/F |
No detrimental effect when sprayed onto excised cucumber leaves |
Jacobson et al. (2001) |
| Aphidius colemani Orius insidiosus Phytoseiulus persimilis Encarsia formosa |
B. Bassiana (commercial formulation, strain JW-1) |
L |
Highly susceptible under laboratory conditions, lower infection rates in greenhouse |
Ludwig and Oetting (2001) |
| Apis mellifera |
B. bassiana |
F |
Conidia were applied in bee hives: low mortality and no noticeable effect on behaviour, larvae and colony characteristics |
Alves et al. (1996) |
| Apis mellifera |
B. bassiana (unformulated spore preparation) |
L |
B. bassiana reduced bee longevity at the two highest concentrations tested and caused mycosis at 106–108 spores per bee |
Vandenberg (1990) |
| Apis mellifera |
B. bassiana (Naturalis-L, Bio-Power) |
L |
30-day dietary and contact studies had no significant effect; LC50 (23 days, ingestion) 9.285 µg/bee |
Copping (2004) |
| Apis mellifera |
B. bassiana |
L |
High mortality in caged bees dusted with dry formulation at high concentration (108-109 CFUg-1)Very low mortality following exposure to high inoculum densities regardless of the isolate. |
Al mazrawi (2007) |
| Apis mellifera |
M. anisopliae, B. bassiana, B. thuringiensis |
L |
M. anisopliae and B. bassiana reduced survival of A. mellifera when sprayed directly, all did not induce morphometric alterations in the midgut. |
Potrich et al. (2017) |
| Arthropod and nematode populations |
B. bassiana (Naturalis-L) |
F |
Chlorpyrifos had a stronger negative impact than the microbial treatment |
Wang et al. (2001) |
| Bembidion lampros Agonum dorsale |
B. bassiana |
F/L |
A negligible number was infected; low susceptibility of both species |
Riedel and Steenberg (1998) |
| Bombus terrestris |
B. bassiana |
L/F |
Able to infect bumblebees; it appears that there are no risks if the fungus is incorporated into the soil or sprayed onto plants that are not attractive to bumblebees |
Hokkanen et al. (2003) |
| Carabidae: Calanthus micropterus C. piceus Carabus violaceus Cychrus caraboides Leistus ruefescens Nebria brevicollis, Pterostichus oblongopunctatus, P. niger |
B. bassiana |
L |
No adverse effects noticed |
Hicks et al. (2001) |
| Carabidae, Staphylinidae |
B. bassiana |
F |
Infection levels in adult ground beetles and rove beetles were low (Carabidae max. 7.6% and Staphylinidae max. 7.0%); an epizootic in the staphylinid Anotylus rugosus (67%) and Gyrohypnus angustatus (37%) was observed |
Steenberg et al. (1995) |
| Cephalonomia tarsalis |
B. bassiana |
– |
3 h exposure to 100 and 500 mg kg−1 wheat resulted in 52.5 and 68.6% mortality |
Lord (2001) |
| Chrysoperla carnea |
B. bassiana |
L |
Temperature, starvation and nutrition stresses significantly affected the susceptibility; nutrition stress caused the most increase in adult and larval mortality |
Donegan and Lighthart (1989) |
| Coleomegilla maculate |
B. bassiana (isolate ARSEF 3113) |
L/F |
No mortality was observed |
Pingel and Lewis (1996) |
| Coleomegilla maculate and Eriopis connexa |
B. bassiana (isolate ARSEF 731) |
L |
Mortality after direct application of spores; exposure via sprayed leaf surfaces resulted in no infection |
Magalhaes et al. (1988) |
| Coleomegilla maculate lengi |
B. bassiana (10 isolates) |
L |
6 isolates were highly virulent, 3 isolates caused low mortality |
Todorova et al. (2000) |
| Diadegma semiclausum |
B. bassiana |
L |
Detrimental effects on cocoon production and emergence depending on the concentration |
Furlong (2004) |
| Formica polyctena |
B. brongniartii |
F |
No negative effects noticed |
Dombrow (1988) |
| Earthworms: Lumbricus terrestris and others |
B. brongniartii (commercial product of barley grains) |
L/F |
No effect in a lab and in field noticed |
Hozzank et al. (2003) |
| Earthworms: Lumbricus terrestris |
B. brongniartii |
L |
No effect on earthworms noticed |
Arregger-Zavadil (1992) |
| Earthworms: Aporrectodea caliginosa |
B. bassiana (Bb64) |
L |
No effect on hatching rate of cocoons |
Nuutinen et al. (1991) |
| Lysiphlebus testaceipe Aphidius colmani |
B. bassiana |
F |
No significant impacts on both parasitoids |
Murphy et al. (1999) |
| Megachile rotundata |
B. bassiana (strain for grasshopper control) |
L |
Spray-application of flowering alfalfa in pots: female and male mortality averaged 9%; no difference in treatment and control; however B. bassiana grew out from dead bees |
Goettel and Johnson (1992) |
| Nontarget arthropods (forests) |
B. brongniartii |
F |
Only 1.1% of 10.165 collected insects and spiders were infected |
Baltensweiler and Cerutti (1986) |
| Nontarget arthropods (forests) |
B. brongniartii |
F |
1671 nontarget specimens were collected: 3.4% of them were infected, mainly species from Araneae, Thysanoptera, Homoptera, Coleoptera and Lepidoptera |
Back et al. (1988) |
| Nontarget arthropods (major predators, parasitoids and pollinators on rangeland) |
B. bassiana (strain GHA) |
F |
No statistical differences in the abundance of aerial insects |
Brinkman and Fuller (1999) |
| Nontarget arthropods (forests) |
B. bassiana (emulsifiable concentrate) |
F |
From 3615 invertebrates collected, only 2.8% became infected; B. bassiana could be applied to forest soil without a significant negative impact on forest-dwelling invertebrate population |
Parker et al. (1997) |
| Non-target beetle communities |
B. bassiana (strain SP 16) |
F |
No detectable effects |
Ivie et al. (2002) |
| Perillus bioculatus |
B. bassiana (six isolates) |
L |
5 isolates were highly pathogenic, isolate IPP46 showed low pathogenicity |
Todorova et al. (2002) |
| Pimelia senegalensis Trachyderma hispida Bracon hebetor Apoanagyrus lopezi |
B. bassiana |
L |
No infection in P. senegalensis and T. hispida; 100% mortality in the parasitoids B. hebetor and A. lopezi |
Danfa et al. (1999) |
| Poecilus versicolor |
B. brongniartii (Melocont-Pilzgerste, Melocont-WP, and Melocont-WG) |
L |
No significant negative effects on P. versicolor could be observed |
Traugott et al. (2005) |
| Predatory mites: O. insidiosus |
B. Bassiana (Botanigard ES) |
F |
Can be used |
Shipp et al. (2003) |
| A. colemani Dacnusa sibiria |
|
|
Not recommended during application of B.bassiana |
|
| Parasites: |
|
|
|
|
| Encarsia formosa Eretmocerus eremicus Aphidoletes aphidimyza |
|
|
Used with caution during application of B. bassiana |
|
| Prorops nasuta |
B. bassiana (3 isolates) |
L |
Strain 25 caused the lowest infection level |
De La Rosa et al. (2000) |
| Serangium parcesetosum |
B. bassiana |
L |
The predator had significantly lower survivorship when sprayed with B. bassiana than with P. fumosoroseus; feeding on B. bassiana contaminated prey caused 86% mortality |
Poprawski et al. (1998) |
Adapted from Zimmermann (2007).
References:
William G. Meikle, Guy Mercadier, Niels Holst, Christian Nansen, Vincent Girod. Impact of a treatment of Beauveria bassiana (Deuteromycota: Hyphomycetes) on honeybee (Apis mellifera) colony health and on Varroa destructor mites (Acari: Varroidae). Apidologie, Springer Verlag, 2008, 39 (2), pp.247-259. Ffhal-00892301f
Marta Rodríguez, Marcos Gerding, Andrés France. Selection of entomopathogenic fungi to control Varroa destructor (Acari: Varroidae). Chilean J. Agric. Res. – Vol. 69 – Nº 4 – 2009
Burges, HD. 1981. “Safety, safety testing and quality control of microbial pesticides”. In Microbial control of pests and plant diseases 1970–1980, Edited by: Burges, HD. 737–767. London: Academic Press.
Hajek, AE and Butler, L. 2000. “Predicting the host range of entomopathogenic fungi”. In Nontarget effects of biological control, Edited by: Follett, PA and Duan, JJ. 263–276. Dordrecht: Kluwer Academic Publishers.
S. Al Mazra’awi, J. L. Shipp, A. B. Broadbent, P. G. Kevan, Dissemination of Beauveria bassianaby Honey Bees (Hymenoptera: Apidae) for Control of Tarnished Plant Bug (Hemiptera: Miridae) on Canola, Environmental Entomology, Volume 35, Issue 6, 1 December 2006, Pages 1569–1577, https://doi.org/10.1093/ee/35.6.1569
Zimmermann, G. (2007) Review on safety of the entomopathogenic fungi Beauveria bassiana and Beauveria brongniartii, Biocontrol Science and Technology, 17:6, 553-596, DOI: 10.1080/09583150701309006
Al Mazra’awi, M. S. (2007). Impact of entomopathogenic fungus Beauveria bassiana on honeybees, Apis mellifera (Hymenoptera: Apidae). Worl Journal of Agricultural Science 3(1): 07-11, 2007.